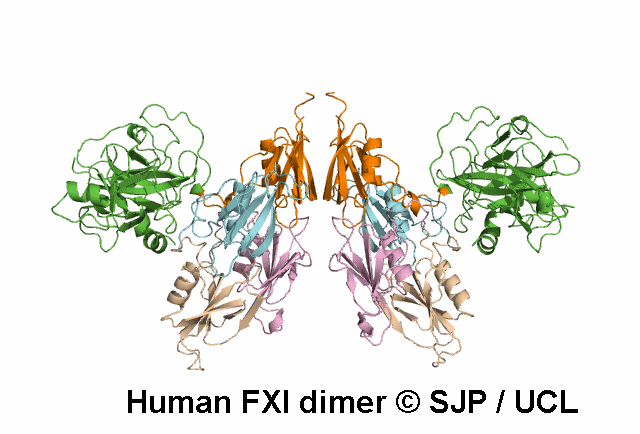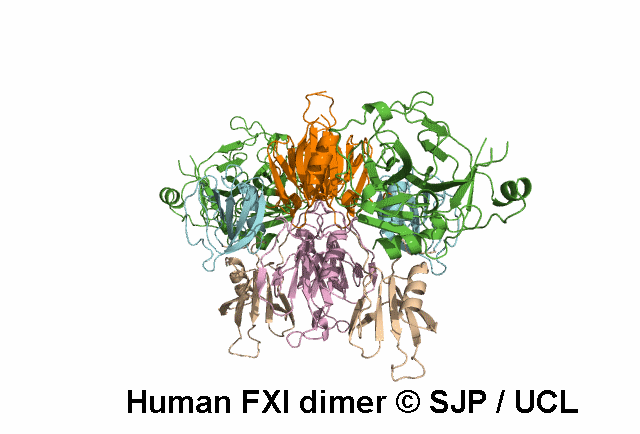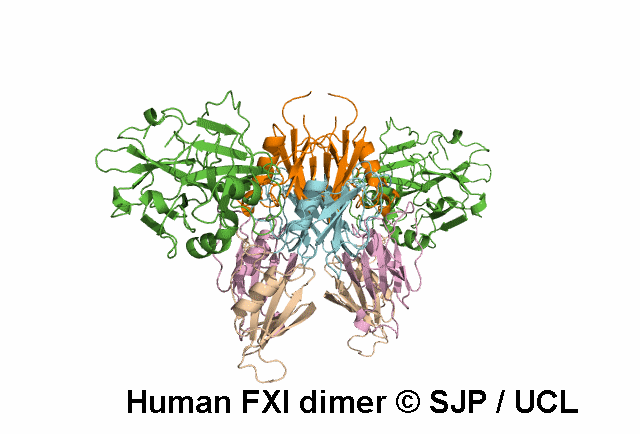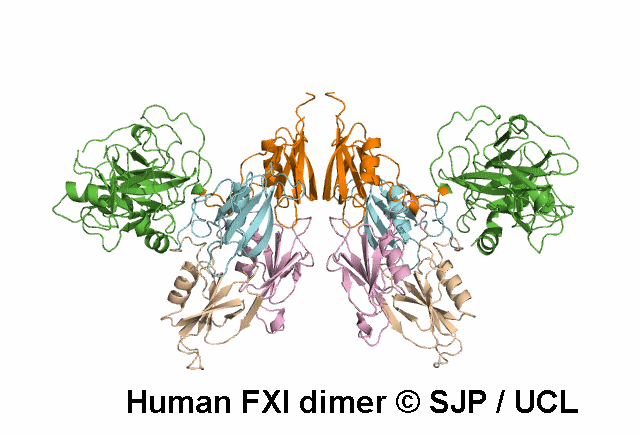In Depth Variant Analysis: c.1789G>A (p.Glu597Lys)
Terms with a '*' next to them are explained on the Help Page .
c.1789G>A
p.Glu597Lys (Legacy AA No. 579)
Variant Type:
Point
Domain:
Serine Protease
Codon Change:
G>A
Variant Effect:
Missense
No. of Patients Reported:
1
Phenotype:
U
Allele Count *:
1
Allele Number *:
251490
Allele Frequency *:
0.000004
Variant Comments & Reference:
Glu597Lys affects overall protein charge. A given patient presented with low levels of FXI activity (15 iu/dL), either due to an undetected mutation in the experimental procedure or due to the fact that Glu597Lys is a dominant mutation that can exert a negative effect on secretion of wild-type FXI. Such dominant mutants have already been described: Trp587Ser, Thr593Met and Ser594Arg. Interestingly, the four mutations are located with in the same 10-amino acid strand, suggesting a critical role for this segment in the dimerisation process. Quelin et al 2006Residue Information:
| Name | Type | Cyclic | Size | Position | Hydrophobicity | Charge | |
|---|---|---|---|---|---|---|---|
| |
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
Substitution Analysis:
Structural Implications:
Glu597 is an exposed residue (surface accessibility value = 5 ).
Glu597 is in a random coil area of the FXI structure.
The DSSP assignment for this residue is ... C.
Glu597 is in a random coil area of the FXI structure.
The DSSP assignment for this residue is ... C.
Hint: Left click mouse | To rotate the structure
Hint: Rotate mousewheel | To zoom in/out the structure
Hint: Right click mouse | to use applet control options
|
| Spacefill | |
| Cartoon | |
| Wireframe | |
| Trace | |
| Backbone | |
| Spin | |
| Background | |
| Disulphides | |
| Domains | |
| Alternative Colouring | |
| Labels |
Right Click on the molecule's screen for more options.
NOTE: If no Java applet appears or a Java error message is shown, please click the following LINK.