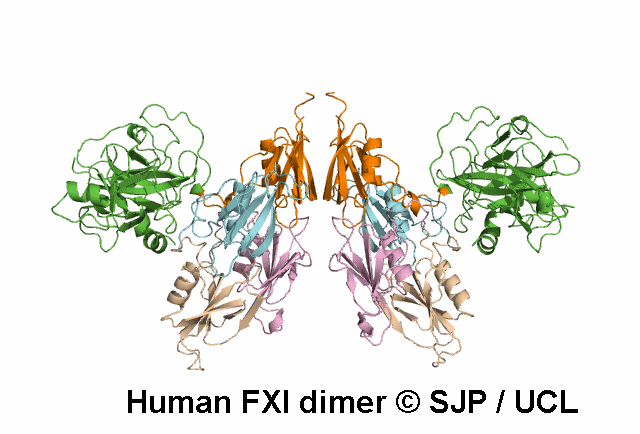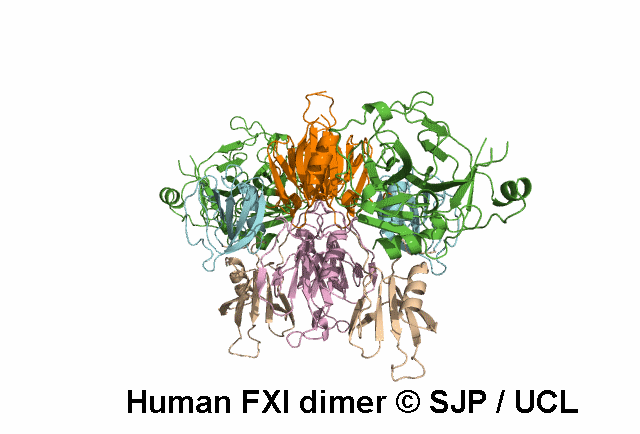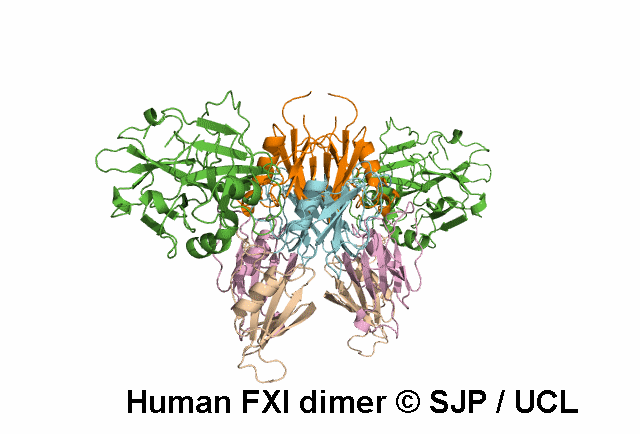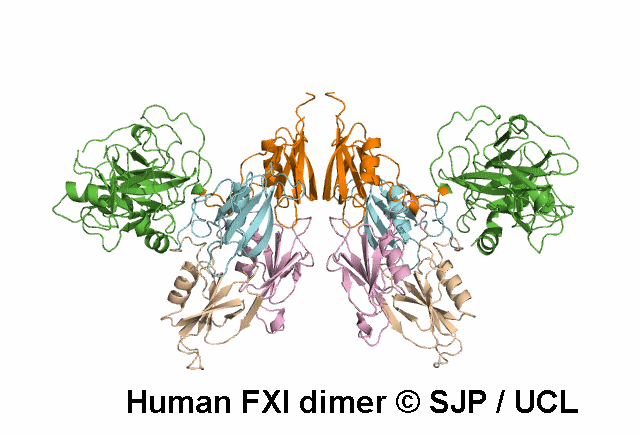In Depth Variant Analysis: c.1718G>A (p.Gly573Glu)
Terms with a '*' next to them are explained on the Help Page .
c.1718G>A
p.Gly573Glu (Legacy AA No. 555)
Variant Type:
Point
Domain:
Serine Protease
Codon Change:
G>A
Variant Effect:
Missense
No. of Patients Reported:
2
Phenotype:
II
Allele Count *:
2
Allele Number *:
251370
Allele Frequency *:
0.000008
Variant Comments & Reference:
Normal amount of mutant protein secreted from BHK cells. Experimental data indicates the mutation changes the conformation of the unoccupied active site of FXIa. When compared with wild type, FXI-Glu573 activates FIX at a greatly reduced rate. Modelling indicates side chain of Glu573 alters electrostatic charge around active site and interferes with the interaction between FXI and FIX and antithrombin. Zivelin et al 2001 (Abstract), Zivelin et al 2004, Schmidt et al 2004Residue Information:
| Name | Type | Cyclic | Size | Position | Hydrophobicity | Charge | |
|---|---|---|---|---|---|---|---|
| |
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
Substitution Analysis:
Structural Implications:
Gly573 is a buried residue (surface accessibility value = 1 ).
Gly573 is in a random coil area of the FXI structure.
The DSSP assignment for this residue is ... C.
Gly573 is in a random coil area of the FXI structure.
The DSSP assignment for this residue is ... C.
Hint: Left click mouse | To rotate the structure
Hint: Rotate mousewheel | To zoom in/out the structure
Hint: Right click mouse | to use applet control options
|
| Spacefill | |
| Cartoon | |
| Wireframe | |
| Trace | |
| Backbone | |
| Spin | |
| Background | |
| Disulphides | |
| Domains | |
| Alternative Colouring | |
| Labels |
Right Click on the molecule's screen for more options.
NOTE: If no Java applet appears or a Java error message is shown, please click the following LINK.